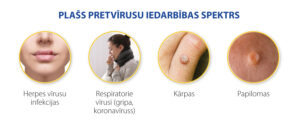
APPLICATION IN PRACTICE dsRNA – LARIFAN
All Larifan brand products contain the active ingredient dsRNA Larifan.
In the status of drugs in its history, several forms of Larifan have been registered:
- injections in Latvia (2004), then in Georgia (2010), Azerbaijan (2011) and Turkmenistan (2015),
- ointment form (0.05%) was registered in Latvia (1994 – 2004), in Georgia (2010)
- suppositories and pessaries in Georgia (2010)
- spray form in Georgia (2010)
In the status of medical goods distributed:
- suppositories and pessaries in Latvia (2008-2011) and Azerbaijan (2010)
- ointment and spray in Azerbaijan (2010)
In the status of cosmetic products:
- lip balm, ointment (0.025%), spray, toothpaste in the European Union (2011) and Georgia (2013)
- lip balm, ointment (0.025%), toothpaste in Moldova (2014) and Vietnam (2016),
- cream for skin inflammation, lotion for sunburn, antibacterial and antiviral gel for hand disinfection – in the territory of the European Union (2016) and Georgia (2017)
- antibacterial and antiviral gel for hand disinfection with natural amber from the Baltic Sea – in the territory of the European Union (2017)
In the status of food products:
- candies against sore throats (5 different flavors) in the European Union (2016) and Georgia (2017)
In Larifan-containing cosmetic and food products, the active substance dsRNA Larifan is included in sub-therapeutic doses, which do not directly treat, but provide the mobilization of the body’s natural defenses, and thus provide some antiviral protection and immunomodulatory effects.